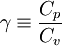Ratio of specific heats
From CFD-Wiki
(Difference between revisions)
Roberthealy1 (Talk | contribs) m (This was well written and informative. However to be truly of encyclopaedic quality references are needed) |
m |
||
| Line 7: | Line 7: | ||
The adiabatic index always exceeds unity; for a [[polytropic gas]] it is constant.{{fact}} For [[monatomic gas]] <math>\gamma=5/3</math>, and for [[diatomic gases]] <math>\gamma=7/5</math>, at ordinary [[temperatures]].{{fact}} For air its value is close to that of a diatomic gas, 7/5 = 1.4.{{fact}} | The adiabatic index always exceeds unity; for a [[polytropic gas]] it is constant.{{fact}} For [[monatomic gas]] <math>\gamma=5/3</math>, and for [[diatomic gases]] <math>\gamma=7/5</math>, at ordinary [[temperatures]].{{fact}} For air its value is close to that of a diatomic gas, 7/5 = 1.4.{{fact}} | ||
| - | Sometimes <math>\kappa</math> is used instead of <math>\gamma</math> to denote the | + | Sometimes <math>k</math> or <math>\kappa</math> is used instead of <math>\gamma</math> to denote the specific heat ratio. |
{{Stub}} | {{Stub}} | ||
Latest revision as of 20:03, 15 August 2007
The ratio of specific heats (also known as adiabatic index), usually denoted by 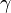 , is the ratio of specific heat at constant pressure to the specific heat at constant volume.
, is the ratio of specific heat at constant pressure to the specific heat at constant volume.
The adiabatic index always exceeds unity; for a polytropic gas it is constant. For monatomic gas 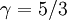 , and for diatomic gases
, and for diatomic gases 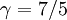 , at ordinary temperatures. For air its value is close to that of a diatomic gas, 7/5 = 1.4.
, at ordinary temperatures. For air its value is close to that of a diatomic gas, 7/5 = 1.4.
Sometimes 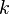 or
or 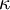 is used instead of
is used instead of 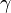 to denote the specific heat ratio.
to denote the specific heat ratio.