Ratio of specific heats
From CFD-Wiki
The ratio of specific heats (also known as adiabatic index), usually denoted by 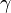 is the ratio of specific heat at constant pressure to the specific heat at constant volume
is the ratio of specific heat at constant pressure to the specific heat at constant volume
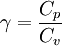
The adiabatic index always exceeds unity; for a polytropic gas it is constant. For monatomic gas 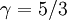 , and for diatomic gases
, and for diatomic gases 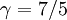 , at ordinary temperatures. For air its value is close to that of a diatomic gas, 7/5.
, at ordinary temperatures. For air its value is close to that of a diatomic gas, 7/5.
