 |
|
|
[Sponsors] | |||||
Species Concentration Initialization for a Methane reduced chemical kinetic mechanism |
 |
|
|
LinkBack | Thread Tools | Search this Thread | Display Modes |
|
|
|
#1 |
|
New Member
Abdelrazik Essam
Join Date: Nov 2018
Posts: 8
Rep Power: 7  |
Hello all,
I'm writing a quasi 1d MATLAB code to simulate pulse jet engine and i'm stuck at the combustion phase. I have to use the reduced 4 reactions-8 species mechanism that was mentioned in "REDUCED REACTION SCHEMES FOR METHANE, METHANOL AND PROPANE FLAMES" by PACZKO. In that mechanism the main species are: CH4,O2,H2,H,H2O,CO,CO2 and M, and to calculate the rates of the reactions i need the concentrations of intermediate species that are OH, O, CHO, HO2, CH3, C2H5, C2H2 and CH. To calculate these intermediates, PACZKO managed to get some algebric equations using the main species for each of them based on some approximations, like steady-state for example (as indicated in the attached 2 images). My question is how to initialize the domain by stoichiometric ratio of fuel/oxidizer and the other species are zeros while they are needed in the denominator of the intermediate species ? |
|
|
|

|
|
|
|
|
#2 |
|
Senior Member
-
Join Date: Jul 2012
Location: Germany
Posts: 184
Rep Power: 14  |
Perhaps you can assume that all species exist up to a small number, e.g.
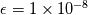 ? ?I have a little trouble to understand your problem. Perhaps you can simplify your question. Or you can use the latex functionality instead of posting some parts of a book. Good questions often correlate with good answers. Regards |
|
|
|

|
|
|
|
|
#3 |
|
New Member
Abdelrazik Essam
Join Date: Nov 2018
Posts: 8
Rep Power: 7  |
Ok, let me simplify this question.
Initially I have the following concentrations for the main species [Kmol/m^3]: C_CH4 = 0.0039 C_O2 = 0.0077 C_M = 0.029 % Third Body treated as Nitrogen C_H2 = C_H = C_H2O = C_CO = C_CO2 = 0 To calculate the rates of reactions for the first time step, I need the concentrations of some intermediate species, let's take C_OH as an example: C_OH = (C_H * C_H2O) / (C_H * Kc3), where Kc3 is a partial-equilibrium constant. So, here C_H, C_H2O and C_H are zeros, then C_OH is undefined number. and some other species tends to infinity because of the existing of ZERO in the denominator. How can I deal with this suitation > |
|
|
|

|
|
|
|
|
#4 |
|
Senior Member
-
Join Date: Jul 2012
Location: Germany
Posts: 184
Rep Power: 14  |
Thank you, now it is easier to follow your problem! However, I think you have a typo in your formulas.
At a first step I would ignore mathematical formulas and consider what physically happens. If only 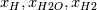 are part to create are part to create 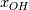 , the answer is zero. , the answer is zero. However if you want a mathematical proof, the rule of L'Hôpital may help you for problems like  at at 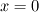 . Here . Here 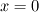 is your concentration. If you assume that all concentration approach with the same speed to zero it holds: is your concentration. If you assume that all concentration approach with the same speed to zero it holds: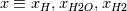 . .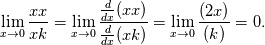 However i do not know if this is the answer to your question. Perhaps you have to solve the problem iteratively if other intermediate chemical reactions create 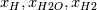 in the meanwhile. in the meanwhile.Regards |
|
|
|

|
|
|
|
|
#5 |
|
New Member
Abdelrazik Essam
Join Date: Nov 2018
Posts: 8
Rep Power: 7  |
Thank you so much for your help. Actually i thought of using L'hopital too, but the other species have nested algebric equations to calculate so this method is complicating.
Another point is, if we think physically, we find it acceptable that these intermediate species have zero concentrations in the beginning, but that assumption will give us zero m_dot fir the main species, i.e, there will be NO change in the initial concentrations and the problem will be the same at the next time step. |
|
|
|

|
|
|
|
|
#6 |
|
Senior Member
-
Join Date: Jul 2012
Location: Germany
Posts: 184
Rep Power: 14  |
||
|
|

|
|
|
|
|
#7 |
|
New Member
Abdelrazik Essam
Join Date: Nov 2018
Posts: 8
Rep Power: 7  |
||
|
|

|
|
|
|
|
#8 |
|
New Member
Abdelrazik Essam
Join Date: Nov 2018
Posts: 8
Rep Power: 7  |
May i ask you another question please that may help us in thinking differently ?
PACZKO mentiones in his paper that for calculating intermediate species, he used partial equilibrium and steady state approximations. But, we know that these approximations relates to fast buil-up then slow consumtion for these species. So, how can we use the final reduced mechanism with these approximations at the beginning for the combustion phase ? |
|
|
|

|
|
|
|
|
#9 |
|
Senior Member
-
Join Date: Jul 2012
Location: Germany
Posts: 184
Rep Power: 14  |
I am not deep in the combustion topic. There may be some other guys here in the forum which are more familiar with chemical reactions.
From my quick point of view you have to solve the whole system iteratively to get the steady state solution for intermediate as also final concentration solutions. |
|
|
|

|
|
|
|
|
#10 |
|
New Member
Abdelrazik Essam
Join Date: Nov 2018
Posts: 8
Rep Power: 7  |
Thank you so much for your effort.
|
|
|
|

|
|
 |
| Tags |
| code, combustion, matlab |
|
|
 Similar Threads
Similar Threads
|
||||
| Thread | Thread Starter | Forum | Replies | Last Post |
| species transport with eulerian multiphase, kinetic theory of granular flow | m.uzair | FLUENT | 4 | October 26, 2018 10:30 |
| Chemical Kinetic Mechanism File Chemkin | SRI28337 | Main CFD Forum | 0 | August 9, 2017 00:59 |
| reduced reaction mechanism for methane combustion using FLUENT | vnm | ANSYS | 1 | August 9, 2016 09:25 |
| using reduced mechanisms in openFOAM | Alish1984 | OpenFOAM Running, Solving & CFD | 4 | August 17, 2012 06:26 |
| Reduced mechanism for Ethylene Combustion | Chaitanya | FLUENT | 1 | May 9, 2011 06:40 |