Heat transfer
From CFD-Wiki
Revision as of 10:52, 2 December 2005 by Sachinshendge (Talk | contribs)
Conduction
Conduction is heat transfer by means of molecular agitation within a material without any motion of the material as a whole. In this mode of heat transfer, heat flow is due to molecular collisions in the substance. Since molecullar collisions increase with temperature (Temperature increases the kinetic energy of the molecules), heat transfer due to conduction increases. Conduction heat transfer through a substance is because of a temperature gradient. The rate of heat transfer by conduction between two regions of a substance is proportional to the temperature difference between them. The constant of propotionality is called thermal conductivity of the material.
Mathematically, it can be described by using the Fourier's law:
Where

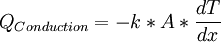
![Q = \mbox{Heat conducted}\;[W]](/W/images/math/c/e/5/ce5124f80c7625563c71a92efbe7c2a6.png)
![k = \mbox{Thermal conductivity of the material}\;[W/m\,K]](/W/images/math/1/f/4/1f4bf7ced6f34761ea9094f175e69505.png)
![A = \mbox{Cross-sectional area of the object perpendicular to heat conduction}\;[m^2]](/W/images/math/e/8/2/e822049913bc6a8f3ae4bdc30e02f2a3.png)
![T = \mbox{Temperature}\;[K]](/W/images/math/2/3/c/23cda486a79eec21f05397bb3942be3e.png)
![x = \mbox{Length of the object}\;[m]](/W/images/math/5/9/a/59a8c70d997eb066da1c3eef42a80b56.png)